2026-04-27
Cool Therapy Pain Relief Patch OEM Manufacturing - Expert Guide 2026
Professional cool therapy pain relief patch OEM manufacturing services. KONGDY Medical offers cooling gel patches with menthol for acute injuries, muscle strain......
Read More
2026-04-27
Heat Therapy Pain Relief Patch OEM Manufacturing - Expert Guide 2026
Professional heat therapy pain relief patch OEM manufacturing services. KONGDY Medical offers warming patches with herbal ingredients for muscle relaxation and ......
Read More
2026-04-24
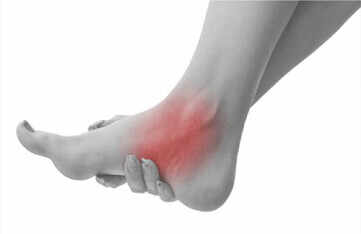
Plantar Fasciitis Pain Relief Patch OEM Manufacturing - Professional Guide 2026
Professional plantar fasciitis pain relief patch OEM manufacturing services. KONGDY Medical offers heel pain relief patches with anti-inflammatory ingredients f......
Read More